Product Overview
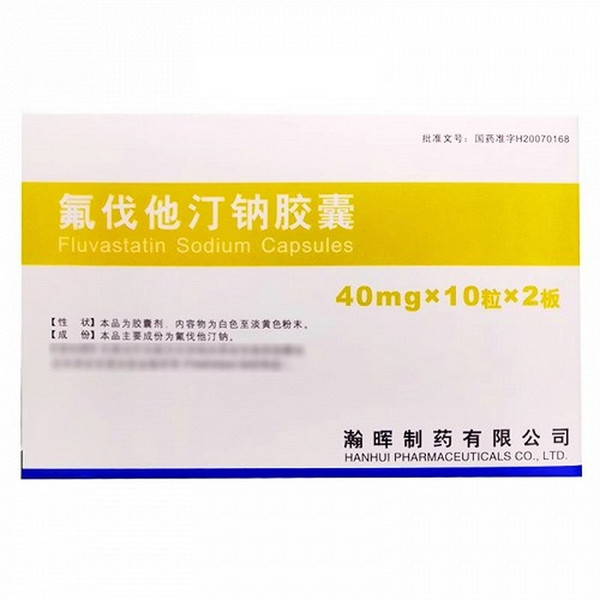
[Drug Name]
Generic Name: Fluvastatin Sodium Capsules
Trade Name: Hisun Fluvastatin Sodium Capsules 40mg x 20 capsules
[Main Ingredient]
Fluvastatin Sodium
[Properties]
This product is in capsule form, containing an off-white to pale yellow powder.
[Indications/Main Functions]
Primary hypercholesterolemia and primary mixed dyslipidemia (Fredrickson types 1a and 1b) not fully controlled by dietary therapy.
[Precautions]
1. Liver Function: As with other cholesterol-lowering medications, liver function tests should be performed before starting Fluvastatin and regularly during treatment. If alanine aminotransferase (AST) or aspartate aminotransferase (ALT) levels are persistently elevated to 3 times or more of the upper normal limit, the drug should be discontinued. There have been isolated reports of possible drug-induced hepatitis. Use with caution in patients with a history of liver disease or those who consume significant amounts of alcohol. 2. Skeletal Muscle Function: Myopathy (including myositis and rhabdomyolysis) has been reported in patients taking other HMG-CoA reductase inhibitors.
[Drug Interactions]
Patients with known hypersensitivity to fluvastatin or any other component of this drug. Active liver disease or persistent unexplained transaminase elevations. Pregnant and lactating women, as well as women of childbearing potential who are not using reliable contraceptive measures. Patients with severe renal insufficiency (creatinine greater than 260 mol/L, creatinine clearance less than 30 ml/min).
[Specifications]
40 mg x 20 tablets
[Dosage and Administration]
Patients must adhere to a low-cholesterol diet before and during treatment with this drug. Usual dose: The recommended dose is 20 or 40 mg once daily. Take with dinner or at bedtime. The dose should be adjusted based on individual response to medication and dietary therapy and recognized treatment guidelines. For patients with very high cholesterol or inadequate response to medication, the dose can be increased to 40 mg twice daily. Maximum low-density lipoprotein cholesterol (LDL-C) lowering is achieved within four weeks of administration. Long-term use provides sustained efficacy. Dosage for Patients with Renal Insufficiency: Because this drug is almost completely eliminated by the liver, with less than 6% excreted in the urine, no dose adjustment is necessary for patients with mild to moderate renal insufficiency. Patients with severe renal insufficiency should not be treated with this drug.
[Adverse Reactions]
1. In placebo-controlled trials, the incidence of the following side effects was greater than 1% in the placebo group: dyspepsia (4.7%), insomnia (1.3%), nausea (1.2%), abdominal pain (1.1%), and headache (1.1%). Dyspepsia was dose-related and was more common in patients receiving the 80 mg/day dose. Adverse reactions with an incidence of 0.5-0.9% were sinusitis (0.9%), flatulence (0.8%), decreased sensation (0.6%), dental problems (0.6%), urinary tract infection (0.6%), and elevated transaminase levels. 2. Since the marketing of fluvastatin sodium, there have been isolated reports of allergic reactions, particularly rash and urticaria. Extremely rare cases have included other skin reactions, thrombocytopenia, angioedema, facial edema, and vasculitis.
[Contraindications]
Patients with known hypersensitivity to fluvastatin or any other component of the drug. Active liver disease or persistent unexplained transaminase elevations. Pregnant and lactating women, as well as women of childbearing potential who are not using reliable contraceptive measures. Patients with severe renal impairment (creatinine greater than 260 mol/L, creatinine clearance less than 30 ml/min).