Product Overview
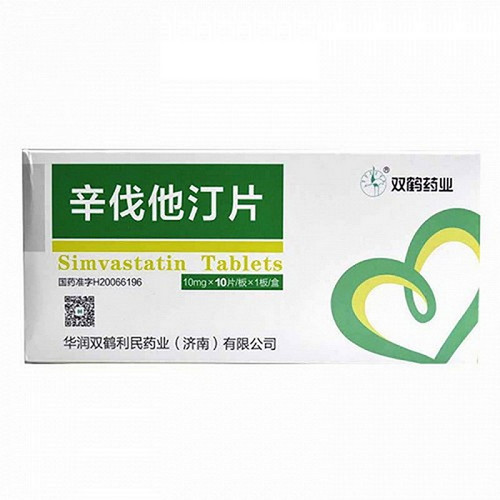
[Drug Name]
Generic Name: Simvastatin Tablets
Trade Name: Sulecon Simvastatin Tablets 10mg x 20 Tablets
[Main Ingredient]
The main ingredient of this product is simvastatin.
[Properties]
This product is a film-coated tablet that appears white or off-white after removal of the coating.
[Indications/Main Functions]
1. Hyperlipidemia. 1) For patients with primary hypercholesterolemia, including heterozygous familial hypercholesterolemia, hyperlipidemia, or mixed hyperlipidemia, when dietary control and other non-drug treatments are unsatisfactory, this product can be used in combination with dietary control to lower elevated total cholesterol, LDL-C, apolipoprotein B, and triglycerides. It can also increase high-density lipoprotein cholesterol, thereby reducing LDL-C, HDL-C, and the total-to-HDL-C ratio. 2) For patients with homozygous familial hypercholesterolemia, this product can be used in combination with dietary control and non-dietary therapies to lower elevated total cholesterol, LDL-C, and apolipoprotein B. 2. Coronary Heart Disease. For patients with coronary heart disease and hypercholesterolemia, this drug is indicated for: 1) reducing the risk of death. 2) reducing the risk of coronary heart disease death and non-fatal myocardial infarction. 3) reducing the risk of stroke and transient ischemic attack. 4) reducing the risk of coronary revascularization surgery (coronary artery bypass grafting and percutaneous transluminal coronary angioplasty) and slowing the progression of coronary atherosclerosis, including reducing the formation of new lesions and total blockages. 3. Children with Heterozygous Familial Hypercholesterolemia. For adolescent boys and girls aged 10-17 years (at least 1 year after menarche) with heterozygous familial hypercholesterolemia, combined with dietary adjustments, this drug can be used to lower total cholesterol, low-density lipoprotein cholesterol, apolipoprotein B, and triglycerides.
[Precautions]
1. Hepatic Reactions: In clinical trials, a small number of patients taking simvastatin experienced significant and persistent elevations in serum aminotransferases (more than three times the normal value). However, after discontinuation of the drug, transaminases return to pre-treatment levels, without jaundice or other concerning clinical signs or symptoms, and without allergic reactions. It is recommended that patients with elevated transaminases before treatment receive increased monitoring and vigilance. If transaminases continue to rise, particularly if they exceed three times the normal value and remain elevated, the drug should be discontinued. This drug should be used with caution in patients who consume significant amounts of alcohol and/or have a history of liver disease. Simvastatin should be contraindicated in patients with active liver disease or unexplained transaminase elevations. As with other lipid-lowering drugs, moderate elevations of transaminases (less than three times the normal value) have been reported in patients treated with simvastatin. These changes typically occur shortly after treatment with simvastatin, are generally transient, and are not associated with any symptoms, so discontinuation of the drug is not necessary. 2. Muscle Reactions: Mild, transient elevations of creatine kinase (CK) (derived from skeletal muscle) are common in patients treated with simvastatin, but these are not clinically significant. Myopathy should be considered in cases of diffuse myalgia, weakness, and/or significant elevations in creatine kinase (CK) (greater than ten times the normal value). Therefore, patients should be advised to report any unexplained myalgia, weakness, or weakness to their doctor immediately. Simvastatin treatment should be discontinued immediately if creatine kinase (CK) is significantly elevated or if myalgia is diagnosed or suspected. Treatment with methylhydroxyglutaryl coenzyme A (HMG-CoA) reductase inhibitors should be discontinued in patients with acute or severe conditions suggestive of myopathy or those at risk for secondary acute renal failure due to rhabdomyolysis. 3. Ophthalmological Examination: The incidence of lens opacities increases with age, even in the absence of any medication. Long-term clinical study data indicate that simvastatin has no adverse effects on the human lens. 4. Use in Pregnancy: No data are available regarding the use of simvastatin in pregnant women. Simvastatin is contraindicated in pregnant women. Because atherosclerosis is a chronic process, discontinuing lipid-lowering medications during pregnancy has little impact on the long-term effectiveness of treating primary hypercholesterolemia. Furthermore, cholesterol and other products of its biosynthetic pathway are essential for fetal development, including the synthesis of steroids and cell membranes. Because HMG-CoA reductase inhibitors such as simvastatin reduce cholesterol synthesis and may also reduce other products of the cholesterol biosynthetic pathway, simvastatin use during pregnancy may be harmful to the fetus. Among women of childbearing age, simvastatin should only be used in those with a low probability of pregnancy. If a woman becomes pregnant while taking simvastatin, simvastatin should be discontinued and the potential for fetal harm should be explained. 5. Breastfeeding Women: It is not known whether simvastatin and its metabolites are excreted in human milk. Because many drugs are excreted in human milk, and given the potential serious side effects of this drug, women taking simvastatin should not breastfeed. 6. Pediatric Use: The safety and efficacy of this drug for pediatric use have not been established. Simvastatin is not currently recommended for use in children. 7. Elderly Use: In controlled clinical trials of simvastatin in elderly patients (over 65 years of age), its efficacy in lowering total cholesterol and low-density lipoprotein (LDL) cholesterol was comparable to that observed in other populations, without a significant increase in the frequency of side effects or laboratory abnormalities. 8. Homozygous Familial Hypercholesterolemia: Because patients with homozygous familial hypercholesterolemia completely lack the low-density lipoprotein (LDL) receptor, simvastatin is less effective in this population. 9. Hypertriglyceridemia: Simvastatin has only a moderate triglyceride-lowering effect and is not suitable for treating conditions characterized by elevated triglycerides (such as Types I, IV, and V hyperlipidemia). Please read the package insert carefully and use as directed by your doctor.
[Drug Interactions]
1. Hypersensitivity to any component of this product. 2. Active liver disease or persistent unexplained elevations in serum transaminases. 3. Pregnant and lactating women (see Precautions, Use During Pregnancy and Lactation).
[Pediatric Use]
The safety and efficacy of simvastatin in patients with heterozygous familial hypercholesterolemia aged 10 to 17 years were evaluated in a controlled trial conducted in adolescent boys and girls (at least 1 year after menarche). Adverse events in patients treated with simvastatin were generally similar to those in the placebo group. Doses greater than 40 mg have not been studied in this population. In this limited controlled study, simvastatin did not show significant effects on growth or sexual maturation in adolescent boys or girls, or on menstrual cycle length in adolescent girls. (See Dosage and Administration; Adverse Reactions; Clinical Trials.) Adolescent girls are advised to use appropriate contraceptive methods during simvastatin treatment. (See Contraindications; Precautions; Use in Pregnant and Lactating Women.) Simvastatin has not been studied in patients younger than 10 years of age or in premenarchal girls.
[Use in Elderly Patients]
In controlled clinical studies of simvastatin in elderly patients (>65 years), its effects on lowering total cholesterol and LDL cholesterol were similar to those seen in other populations, with no significant increase in adverse reactions or laboratory abnormalities.
[Use in Pregnant and Lactating Women]
1. Pregnant Women. This product is contraindicated in pregnant women. There are no safety data on simvastatin use in pregnant women. No controlled clinical trials of simvastatin have been conducted in pregnant women. There are rare reports of birth defects associated with HMG-CoA use during pregnancy. However, a retrospective analysis of approximately 200 patients who used simvastatin or other closely related HMG-CoA inhibitors during the first trimester of pregnancy found a birth defect rate similar to that in the general population. This number of patients was statistically sufficient to rule out a birth defect rate that was 2.5 times or more higher than the general population. Although there is no definitive evidence that simvastatin use in pregnant women increases the risk of birth defects, simvastatin can reduce fetal mevalonate (a precursor for cholesterol biosynthesis). Atherosclerosis is a chronic process, so discontinuing lipid-lowering medications during pregnancy has minimal impact on the long-term effectiveness of treating primary hypercholesterolemia. Therefore, this drug is contraindicated in pregnant women, women who are planning to become pregnant, or women who may become pregnant. Use of this drug should be discontinued during pregnancy (see Contraindications). 2. Breastfeeding Women. It is not known whether simvastatin and its metabolites are excreted in human milk. Because many drugs are excreted in human milk and may cause serious adverse reactions, women taking this drug should not breastfeed (see Contraindications).
[Dosage and Administration]
Patients should follow a standard cholesterol-lowering diet before treatment with this drug and continue to do so during treatment. The recommended starting dose is 20 mg daily, taken once in the evening. For patients requiring only a moderate reduction in LDL cholesterol, the starting dose is 10 mg. For patients taking concomitant cyclosporine, danazol, fibrates (except fenofibrate) or niacin, amiodarone, verapamil, and those with severe renal insufficiency, the recommended doses are as follows. The recommended dosage range is 5-80 mg per day, taken once in the evening. The dosage should be individually adjusted based on baseline LDL cholesterol levels, recommended treatment goals, and patient response. Dose adjustments should be made at intervals of 4 weeks or more. Cholesterol levels should be monitored regularly, and a dose reduction should be considered if the LDL cholesterol level drops below 75 mg/dL (1.94 mmol/L) or the total plasma cholesterol level drops below 140 mg/dL (3.6 mmol/L). -- Homozygous Familial Hypercholesterolemia: Based on the results of a controlled clinical study, the recommended dosage of this drug for patients with homozygous familial hypercholesterolemia is 40 mg per day, taken once in the evening, or 80 mg per day, divided into 20 mg, 20 mg, and 40 mg in the evening. This drug can be used in combination with other lipid-lowering therapies (such as LDL aspiration) or alone when these methods are unavailable.
[Adverse Reactions]
1. This drug is generally well tolerated, with most adverse reactions being mild and transient. In controlled clinical studies, less than 2% of patients discontinued treatment due to adverse reactions. In premarketing controlled clinical studies, adverse reactions considered by investigators to be drug-related (classified as possible, probably, or definitely) with an incidence of ≥1% included abdominal pain, constipation, and flatulence; and fatigue and headache, with an incidence of 0.5-0.9%. Myopathy was reported rarely. In the HPS study (see Clinical Trials), 20,536 patients received either 40 mg of simvastatin (n=10,269) or placebo (n=10,267) daily for a mean observation period of 5 years. The safety profile was similar between the two groups. This large trial only recorded serious adverse reactions and withdrawals due to side effects. Discontinuation rates due to adverse reactions were similar between the two groups (4.8% in the simvastatin group and 5.1% in the placebo group). The incidence of myopathy in the simvastatin group was less than 0.1%. Elevated transaminases (≥3 times the upper limit of normal on repeated examinations) occurred in 0.21% (n=21) of the simvastatin group and 0.09% (n=9) of the placebo group, respectively. 3. In the Nordic Simvastatin Survival Study (4S), 4444 patients received 20-40 mg of simvastatin daily (n=2221) or placebo (n=2223) daily for a median follow-up of 5.4 years. Safety and tolerability were similar between the two groups. 4. Reported in uncontrolled clinical studies or post-marketing use.
[Contraindications]
1. Hypersensitivity to any component of this product. 2. Active liver disease or persistently elevated serum transaminases. 3. Pregnant and lactating women (see Precautions, Use in Pregnancy and Lactation).
[Overdose]
Rare reports of overdose have occurred; the maximum dose was 3.6 g. All patients recovered without sequelae. Overdose is generally managed with standard measures.
[Pharmacology and Toxicology]
Simvastatin can lower normal and elevated low-density lipoprotein cholesterol (LDL-C) concentrations. LDL is formed from very low-density lipoprotein (VLDL) and is primarily catabolized by the high-affinity LDL receptor. The mechanisms by which simvastatin lowers LDL-C include: reducing VLDL-cholesterol concentrations, inducing LDL receptors, leading to a decrease in LDL cholesterol, and increasing LDL-C catabolism. Apolipoprotein B (apo B) levels also decrease significantly during simvastatin treatment. Since each LDL particle contains one molecule of apo B and apo B is rarely found in other lipoproteins, this suggests that simvastatin not only removes cholesterol from LDL but also reduces the concentration of circulating LDL particles. Furthermore, simvastatin can increase high-density lipoprotein cholesterol (HDL-C) concentrations and reduce plasma triglycerides (TG). These factors can lead to reductions in total cholesterol/HDL-C and LDL-C/HDL-C ratios.
[Pharmacokinetics]
Following oral administration, simvastatin is highly selective for the liver, with liver concentrations significantly higher than in other non-target tissues. The majority of simvastatin undergoes extensive first-pass absorption in the liver, acting primarily there and subsequently excreted via the bile. Less than 5% of the active simvastatin fraction is found in the periphery, and 95% of this is bound to plasma proteins. The specific metabolic pathway of fusidic acid in the liver is unknown, but interactions between fusidic acid and HMG-CoA reductase inhibitors metabolized by CYP-3A4 are suspected.